The battery is the main source of electrical energy in our vehicles. It stores chemicals, not electricity. Two different types of lead in an acid mixture react to produce an electrical force. This electrochemical reaction changes chemical energy to electrical energy. The battery powers the following major electrical systems:
- Starting
- Ignition
- Charging
- Lighting (in bikes without starting the engine)
- Accessories
A storage battery is an electrochemical device that produces voltage and delivers electrical current. The battery is the primary “source” of electrical energy used in vehicles today. It is important to remember that a battery does not store electricity, But rather it stores a series of chemicals, and through a chemical process electricity is produced.
The main functions of battery are:
- Engine Off: Electricity from the battery is used to operate lighting, accessories, or other electrical systems when the engine is not running.
- Engine Starting: Current from the battery is used to operate the starter motor in electric start vehicles and to provide sufficient current for the ignition system during engine cranking.
- Engine Running: Electricity from the battery may be needed to supplement the charging system when the vehicle’s electrical load requirements exceed the charging system’s ability to produce electricity. Both the battery and the alternator supply electricity when demand is high. Generally, this circumstance doesn’t crop up in two wheelers as we have very less electrical stuff.
Battery Construction
A common type of lead acid battery contains case, plates, separators, venting system and electrolyte.
Battery Case
Case is a container which holds and protects all battery components and electrolyte, separates cells, and provides space at the bottom for residue (active materials washed off plates). Semi-transparent plastic cases allow checking electrolyte level without removing vent caps.
Cover is permanently sealed to the top of the case; provides outlets for positive and negative terminal posts, vent holes for venting of gases and for battery maintenance (checking electrolyte, adding water). The battery case and cover.
- Form a sealed container
- Protect the internal parts
- Keep the internal parts in proper alignment
- Prevent electrolyte leakage
Plates
Positive and negative plates have a grid framework of antimony and lead alloy. Active material is pasted to the grid: brown-colored lead dioxide (PbO2) on positive plates, gray-colored sponge lead (Pb) on negative plates. The number and size of the plates determine current capability of the battery. Batteries with large plates or many plates produce more current than batteries with small plates or few plates.
Two types of plates are used in a batteries: positive and negative.
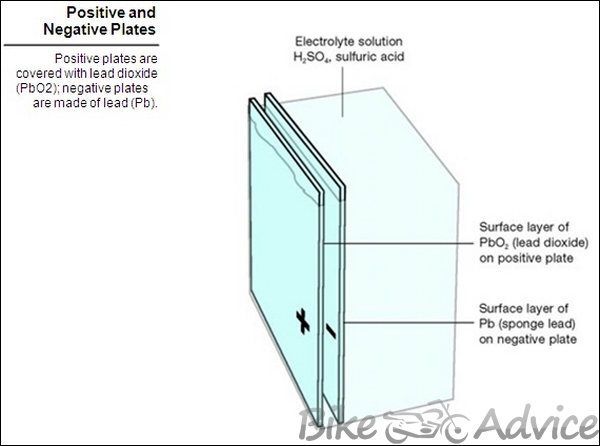
Positive − Positive plates are made of antimony covered with an active layer of lead dioxide (PbO2).
Negative − Negative plates are made of lead covered with an active layer of sponge lead (Pb).
Only the surface layers on both plates take part in the chemical reaction.
Plate surface area − As the surface area of the plates increases, so does the current capacity of the battery. Surface area is determined by the size of each plate, as well as the total number of plates in a battery. Generally speaking, the larger the battery, the higher is its current capacity.
Separators
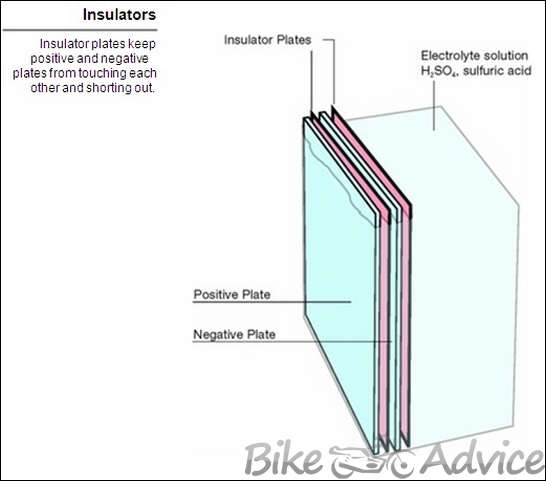
Separators are thin, porous insulators (woven glass or plastic envelopes) that are placed between positive and negative plates. They allow passage of electrolyte, yet prevent the plates from touching and shorting out. These also allow electrolyte to pass freely between the plates.
Cells
An assembly of connected positive and negative plates with separators in between is called a cell or element. When immersed in electrolyte, a cell produces about 2.1 volts (regardless of the number or size of plates). Battery cells are connected in series, so the number of cells determines the battery voltage. A “12 – volt” battery has six cells. Each cell
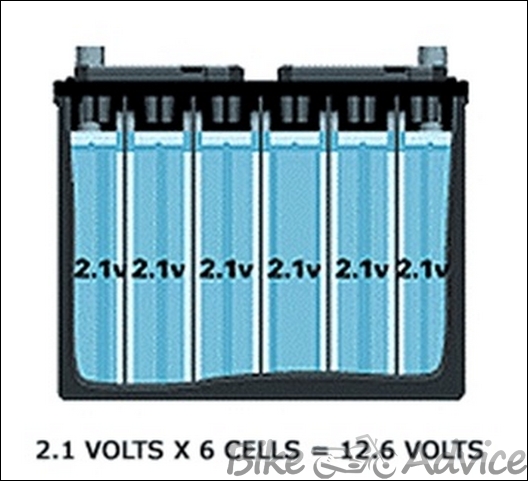
- Consists of multiple positive and negative plates immersed in their own electrolyte reservoir
- Produces about 2.1 volts, regardless of battery size.
Two wheeler batteries were rated at 6 volts and nowadays 12 volts. To make up this voltage, six cells, each producing 2.1 volts, are connected in series.
Venting System
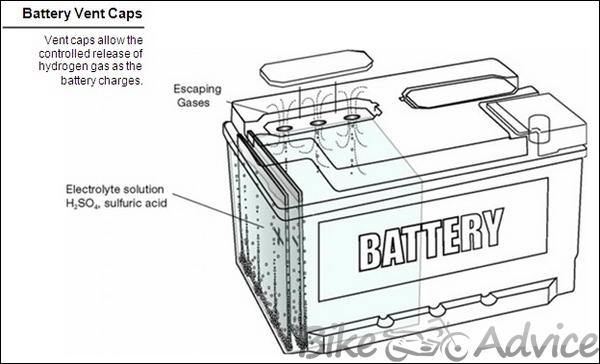
On some batteries, vent caps allow a controlled release of hydrogen gas. This gas forms naturally during battery recharging, whether by the vehicle’s alternator or by an external charger. This includes individual filler plugs, strip-type, or box-type. On removing the plugs, one can check the electrolyte and, if necessary, can add distilled water.
Electrolyte
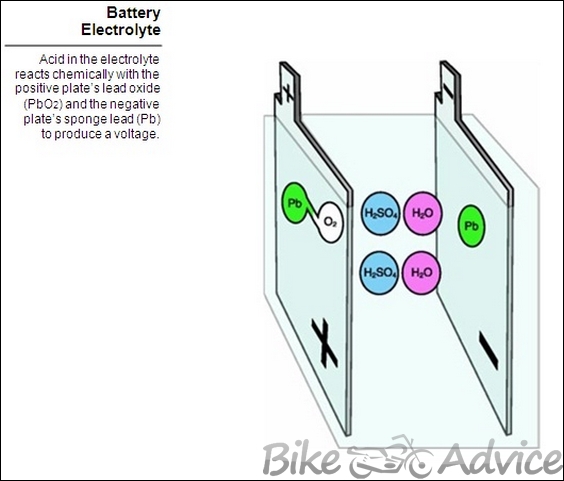
The electrolyte is a mixture of sulfuric acid (H2SO4) and water (H2O). The electrolyte reacts chemically with the active material on the plates to produce a voltage (electrical pressure). And, it conducts the electrical current produced by that pressure from plate to plate. A fully charged battery will have about 36% acid and 64% water.
Capacity Ratings
The battery must be capable of cranking the engine and providing adequate reserve capacity. Its capacity is the amount of electrical energy the battery can deliver when fully charged. Capacity is determined by the size and number of plates, the number of cells, and the strength and volume of electrolyte. In India, we are having AH rating systems for vehicle batteries.
AMP-Hours (AH)
The battery must maintain active materials on its plates and adequate lasting power under light-load conditions. This method of rating batteries is also called the 20-hour discharge rating. Original equipment batteries are rated in amp-hours. The ratings of these batteries are listed on the specifications. The Amp-Hour Rating specifies, in amp hours, the current the battery can provide for 20 hours at 80˚F (26.7˚C) while maintaining a voltage of at least 1.75 volts per cell (10.5 volts total for a 12-volt battery). For example, a battery that can deliver 1 amp for 20 hours is rated at 20 amp-hours (1 x 20 = 80).
Battery Testing
Battery testing has changed in recent years; although the three areas are basically the same.
- Visual Inspection
- State of Charge
- Specific Gravity
- Open Circuit Voltage
- Capacity or Heavy Load Test
As we generally don’t have equipment to conduct the second and third types of tests, atleast we can do the visual inspection.
Visual inspection
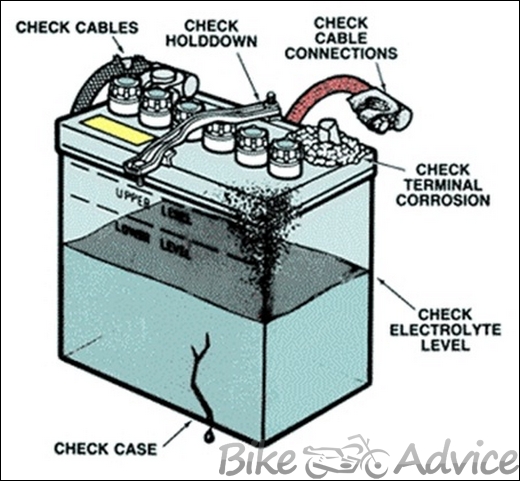
Battery service should begin with a thorough visual inspection. This inspection may reveal simple, easily corrected problems.
- Check for cracks in the battery case and broken terminals. Either may allow electrolyte leakage, which requires battery replacement.
- Check for cracked or broken cables or connections. Replace, as needed, because broken cables can create unnecessary body earth.
- Check for corrosion on terminals and dirt or acid on the case top. Clean the terminals and case top with a mixture of water and baking soda. A wire brush tool is needed for heavy corrosion on the terminals.
- Check for a loose battery hold-down or loose cable connections. Clean and tighten, as needed.
- Check the electrolyte fluid level. The level can be viewed through the semi-transparent plastic case or by removing the vent caps and looking directly into each cell. The proper level is 1/2″ above the separators (about 1/8″ below the fill ring shown below). Add distilled water if necessary. Do not overfill.
- Check for cloudy or discolored electrolyte caused by overcharging or vibration. This could cause high self discharge. Correct the cause and replace the battery.
8 Basic Tips on How to Ride a New Bike
Causes Of Battery Failure
Corrosion: Spilled electrolyte and condensation from gassing may cause corrosion on terminals, connectors and metal hold downs. Such corrosion increases electrical resistance which reduces available voltage and charging effectiveness. It may also create a current leakage path to allow self-discharge.
Electrolyte Level: A low level exposes active material, and any sulphate hardens and resists chemical action. Loss of electrolyte may be caused by a cracked case, poor maintenance or severe overcharging which causes high internal heat and excessive gassing. Too much electrolyte is just as bad. Overfilling dilutes the electrolyte and spillage may corrode battery terminals.
Overcharging: Overcharging causes excessive gassing and high internal heat. Too much gassing can wash active materials off the plates, as well as cause excessive water usage. Too much heat can oxidize the positive plate material and wrap the plates.
10 Tips to Avoid Accidents While Riding in City: Most Basic But Often Neglected
Undercharging: A faulty charging systems will not maintain the battery at full charge. Severe undercharging allows sulphate on the plates to become hard and impossible to remove by normal charging. The weak electrolyte freezes easier. The undercharging battery may fail to crank the engine.
Vibration: A battery must be mounted securely. Vibration can loosen connections, crack the case, and damage internal components.
Anti-Lock Braking System (ABS) in Motorcycles – Explained
We now already get maintenance free batteries in Indian. These batteries are bit costlier and last longer than the conventional batteries.
– Dhruv Panchal




